Greetings Hive
There has been a lot of fuss/informationwar in the last month
about Graphene Oxide in vaccines. Spanish researchers
allegedly found GO or something similar in the Pfizer vaccine.
In this world anybody can "fake" any news, a sign of truth is
when different trustworthy sources with different specialists keep
finding the same or similar. Then you are coming closer to the truth.
So unless you don't have an independent laboratory testing the
Covid19-vaccinated people & the Covid19-vaccines (randomly),
we can "only" look at the patents, that do exist.
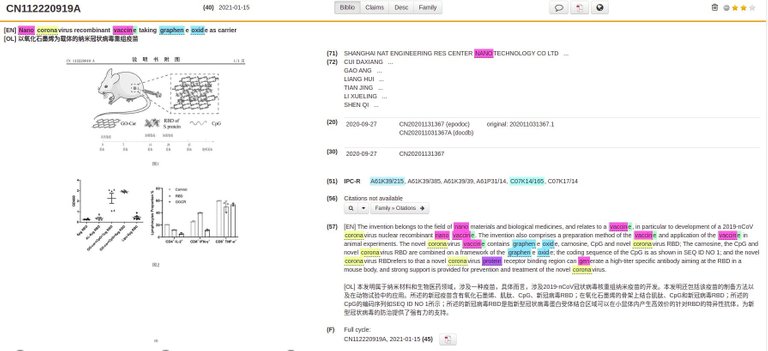
Chinese Patent CN112220919A:
Claims [EN]
1.A coronavirus vaccine, wherein said coronavirus comprises graphene oxide, carnosine, CpG, and a novel coronavirus receptor binding domain; binding carnosine, CpG, and a novel coronavirus receptor binding region on a backbone of graphene oxide; the CpG coding sequence is shown as SEQ ID NO 1; the novel coronavirus receptor binding region refers to a novel coronavirus S protein receptor binding region.
2.The coronavirus of claim 1, wherein the coronavirus is obtained by linking carnosine, CpG and a coronavirus receptor binding region on activated graphene oxide.
3.The method of producing a coronavirus according to claim 1, comprising the steps of:
obtaining CpG, a recombinant protein of a receptor binding region and carnosine, wherein the CpG coding sequence is shown as SEQ ID NO 1;
adding the graphene oxide freeze-dried powder into a phosphate buffer solution, and carrying out ultrasonic treatment;
adding EDC and NHS to activate the graphene oxide solution, removing excessive EDC/sulfo-NHS in the reaction solution through ultrafiltration, and adjusting the pH of the reaction solution to be neutral;
adding carnosine, CpG, and receptor binding region recombinant protein to a reaction solution, and incubating with activated graphene oxide;
excess unconjugated protein was removed from the reaction solution and sterilized for use.
4.The method of claim 3, wherein the duration of sonication is between 2 and 3 hours.
5.The method according to claim 3, wherein the phosphate buffer has a pH of 6.8 to 7.6.
6.The method of claim 3, wherein the method of removing excess EDC/sulfo-NHS or removing uncoupled proteins is ultrafiltration.
7.The method according to claim 3, wherein the amount of carnosine is 1.5 times or more the amount of graphene oxide, the amount of the receptor binding region is 2-10 times the amount of CpG, and the amount of CpG is one ten thousandth of graphene oxide, based on mass ratio.
8.The process according to any one of claims 3 to 7, wherein the reaction temperature is 20 to 28 ℃.
9.Use of a coronavirus according to claim 1, characterised in that said new corona vaccine is used for the preparation of a medicament for the prevention of a coronavirus.
10.The use of claim 9, wherein the coronavirus causes the body to produce antibodies to recombinant proteins of the receptor binding domain.
Abstract:
[EN] The invention belongs to the field of nano materials and biological medicines, and relates to a vaccine, in particular to development of a 2019-nCoV coronavirus nuclear recombinant nano vaccine. The invention also comprises a preparation method of the vaccine and application of the vaccine in animal experiments. The novel coronavirus vaccine contains graphene oxide, carnosine, CpG and novel coronavirus RBD; The carnosine, the CpG and novel coronavirus RBD are combined on a framework of the graphene oxide; the coding sequence of the CpG is as shown in SEQ ID NO 1; and the novel coronavirus RBDrefers to that a novel coronavirus protein receptor binding region can generate a high-titer specific antibody aiming at the RBD in a mouse body, and strong support is provided for prevention and treatment of the novel coronavirus.
[OL] 本发明属于纳米材料和生物医药领域,涉及一种疫苗,具体而言,涉及2019‑nCoV冠状病毒核重组纳米疫苗的开发。本发明还包括该疫苗的制备方法以及在动物试验中的应用。所述的新冠疫苗含有氧化石墨烯、肌肽、CpG、新冠病毒RBD;在氧化石墨烯的骨架上结合肌肽、CpG和新冠病毒RBD;所述的CpG的编码序列如SEQ ID NO 1所示;所述的新冠病毒RBD是指新型冠状病毒蛋白受体结合区域可以在小鼠体内产生高效价的针对RBD的特异性抗体,为新型冠状病毒的防治提供了强有力的支持。
Chinese Patent CN112089834A:
Claims [EN]
1.The pachyman nano adjuvant based on the graphene oxide material is characterized in that a carrier material of the pachyman nano adjuvant is nano graphene oxide, and pachyman is connected to a nano graphene oxide carrier to form the pachyman nano adjuvant based on the graphene oxide material.
2.The pachyman nano adjuvant based on graphene oxide material according to claim 1, characterized in that the graphene oxide material is nano-processed nano-graphene oxide, the particle size of the nano-graphene oxide is 1-100 nm, and the molecular weight is 5-10 kDa; the molecular weight of the pachyman is 8-15 kDa, the constitutional monosaccharides of the pachyman comprise glucose, mannose, fucose, galactose and the like, and the mass ratio of the constitutional monosaccharides is (2-3.5): (1-2): (0.3-4): 1.
3.The pachyman nano adjuvant based on graphene oxide materials as claimed in claim 1, wherein the mass ratio of nano graphene oxide to pachyman is 1: 2.5-1: 10.
4.The pachyman nano-adjuvant based on graphene oxide material as claimed in claim 1, wherein the particle size of the pachyman nano-adjuvant is 50 nm-500 nm, and the molecular weight is 15 kDa-45 kDa.
5.The method for preparing the pachyman nano adjuvant based on graphene oxide material as claimed in claim 1, is characterized by comprising the following steps:
(1) dissolving graphene oxide in sterile water, carrying out ultrasonic treatment for 2 hours, carrying out ice bath ultrasonic treatment for 30 minutes, adding strong base, and carrying out ultrasonic treatment for 2 hours; centrifuging at a high speed of 16,000g, and collecting supernatant to obtain a nano graphene oxide carrier solution, wherein the final concentration of the nano graphene oxide carrier solution is 0.1-10 mg/mL;
(2) carrying out ultrasonic treatment on the nano graphene oxide solution in the step (1) for 1 hour, adjusting the pH value to 9-10, adding epoxy chloropropane, introducing nitrogen for reaction, stirring and reacting at the temperature of 40 ℃ in a water bath for 4 hours, and dialyzing to remove unreacted epoxy chloropropane;
(3) and (3) dissolving the pachymaran solution in the nano graphene oxide solution in the step (2), adjusting the pH value to 9-10, carrying out water bath at 42 ℃ for 3 hours, and centrifuging and collecting to obtain the pachymaran nano adjuvant coated with nano graphene oxide.
6.An adjuvant/antigen co-delivery vaccine comprising the pachyman nano-adjuvant of claim 1 and loaded with a viral antigen.
7.An adjuvant/antigen co-delivery vaccine according to claim 6, wherein the viral antigens comprise: inactivating the virus or recombinant protein.
8.The adjuvant/antigen co-delivery vaccine of claim 6, wherein the antigen and pachyman of the adjuvant/antigen co-delivery vaccine are both linked to graphene oxide.
9.A method of preparing an adjuvant/antigen co-delivery vaccine according to claim 6, comprising the steps of:
(1) adjusting the pH value of the pachymaran nano adjuvant solution encapsulating the nano graphene oxide to 4.5-7.2, adding EDC (1- (3-dimethylaminopropyl) -3-ethylcarbodiimide hydrochloride) and Sulfo-NHS (N-hydroxythiosuccinimide), mixing for 15 min-1 h, adjusting the pH value to 6-8, adding a virus antigen solution, mixing for 1-5 h, and desalting by using a desalting column to obtain the adjuvant/antigen co-delivered vaccine;
EDC/Sulfo-NHS molar ratio is 1: 2-10: 1, the concentration of the virus antigen solution is 0.1-20 mg/mL; the EDC/graphene oxide concentration ratio is 1: 5-1: 20, preferably 1: 10.
10.use of a pachyman nanoadjuvant or an adjuvant/antigen co-delivery vaccine formed from the adjuvant according to any one of claims 1 to 9, for the field of prophylactic and therapeutic vaccines.
.....
[0006] Nanomaterial refers to single crystals or polycrystalline bodies with grain size less than 100 nm. The nanoparticles have biological characteristics of being easily taken up by various cells, and can be phagocytized by antigen presenting cells, so that the immune response caused by antigens is enhanced. The nano material can also slowly release the antigen, reduce the dosage or times of the antigen, increase the size of the small molecule antigen, and fully process and treat the antigen, so that the immune effect is more durable. Some nanoparticles themselves have a stimulatory effect on the immune system and immunization with vaccines containing nanomaterials can result in an immune response in the mucosal and gastrointestinal mucosa. Graphene is a carbon nano-material with a two-dimensional plane structure, and the special monoatomic layer structure of the graphene enables the graphene to have a plurality of unique physicochemical properties. Graphene oxide is an oxide of graphene, and since graphene oxide contains more oxygen-containing functional groups after oxidation, properties of graphene oxide can be improved by various reactions with the oxygen-containing functional groups. The graphene oxide and the derivatives thereof have the advantages of high specific surface area, strong electric and thermal conductivity, good biocompatibility and the like, and are widely applied to the biomedical fields of drug carriers, cancer detection and treatment and the like. Therefore, pachyman is needed to be selected as a raw material, a graphene oxide coating method is adopted, the medicine is slowly released, the dosage of the pachyman is reduced, and an effective, safe and stable immunopotentiator, namely the pachyman nano adjuvant based on the graphene oxide material, is found.
Both patents show GO as a carrier for an so called "adjuvant" to boost immune response, but the most interesting hint i found in the description of the background knowledege:
!...The graphene oxide and the derivatives thereof have the advantages of high specific surface area, strong electric and thermal conductivity, good biocompatibility and the like,....!

Graphene, a promising transparent conductor
So why would you try to raise conductivity in humans by injecting them vaccines with GO? Tracking & Control?
If it's all conspiracy, why do they mention it in the introduction of the patent?😏
Could there be anything positive about a raised conductivity in an organic human body!?
I doubt.
The applicants "SHANGHAI NAT ENGINEERING RES CENTER NANOTECHNOLOGY CO LTD" or
"UNIV BEIJING TECHNOLOGY" most probably want to make money with their "inventions"/
"use" them.
They filed the patents in Sept. & October 2020. They are "pending" right now....
Why wouldn't they "use" their "inventions" if they get the patents granted?
The "technology" is there, how real it is in the Covid19 vaccines can only
be proven by independent laboratories/doctors like i mentioned in the beginning.
I hope this brings us closer to the truth...the One and only Truth.
Stay safe & God Bless
Patent Research
Congratulations @patentresearch! You have completed the following achievement on the Hive blockchain and have been rewarded with new badge(s) :
Your next target is to reach 300 upvotes.
You can view your badges on your board and compare yourself to others in the Ranking
If you no longer want to receive notifications, reply to this comment with the word
STOP